Get our latest news, offers and updates delivered to your inbox

Helping improve animal health and well-being is at the heart of who we are and what we do.
We are dedicated to those who care for animals, whether on the farm, in clinics, or in their homes.

You are the future.
And, it never looked brighter.
We are proud to support Canadian veterinary students with scholarships, immersive learning opportunities, mental health resources and more.

Let’s make the world a better place.
For animals, for people, for our industry, for the environment.
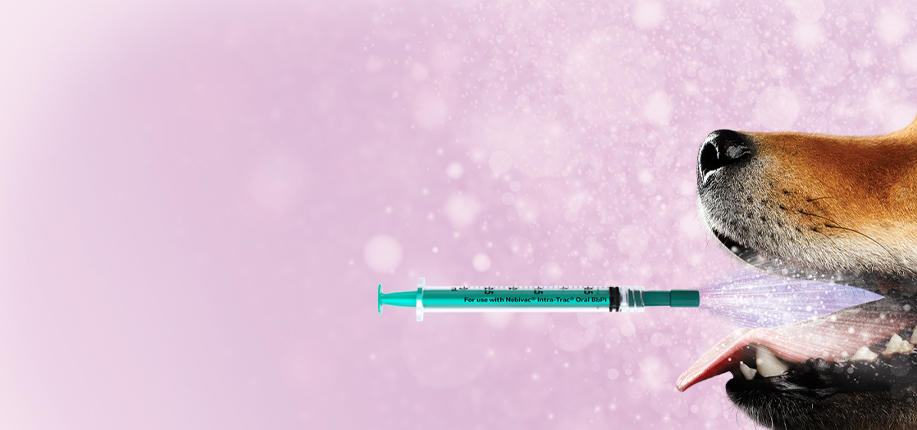

Twist, Mist & Go!
An oral vaccine against Bordetella bronchiseptica and parainfluenza virus has never been possible… until now.
For illustration purposes only. Refer to product insert for complete administration instructions.
NOBIVAC® and INTRA-TRAC® are registered trademarks of Intervet International B.V. All trademarks are used under license.
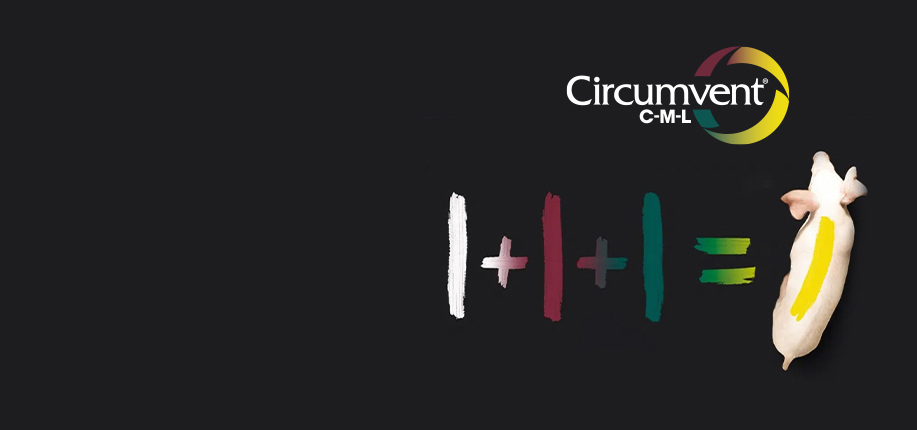
First of its kind
Our latest innovation in swine vaccines provides protection against porcine circovirus type 2, Mycoplasma hyopneumoniae and Lawsonia intracellularis in just one dose.
CIRCUMVENT® is a registered trademark of Intervet International B.V. Used under license.

